Advanced LiFePO₄ (Lithium Iron Phosphate) Powder for Li-ion Battery Cathodes
Lithium iron phosphate (LiFePO₄, or LFP) has become a dominant cathode material in modern lithium-ion batteries, particularly in electric vehicles (EVs), grid-scale energy storage, and high-reliability industrial applications.
While its inherent thermal stability and safety are well recognized, the performance of LFP-based batteries hinges critically on the quality and engineering of the underlying powder. Recent advances in particle morphology control, surface modification, and electronic conductivity enhancements are now pushing LFP performance to match or even surpass some nickel- or cobalt-based cathodes in practical applications.
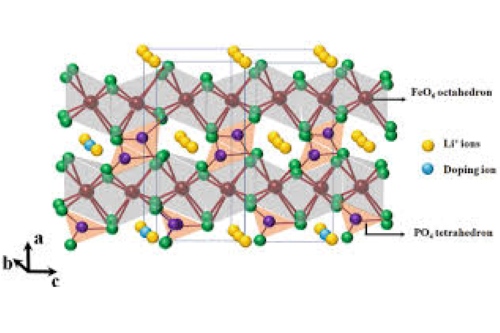 [1]
[1]
Particle Morphology and Its Role in Performance
The electrochemical characteristics of LFP are significantly affected by powder morphology. The perfect morphology of an LFP particle should have a small particle size, good uniformity, and high tap density. This ensures that the electrodes are packed tightly, allowing good diffusion for lithium ions. The particle diameter should range from 100 nanometers to 5 microns. The best powders, especially those used for high-rate discharge, should be nanoscale, as this minimizes internal resistance.
Hydrothermal synthesis, solvothermal synthesis, and spray pyrolysis are new technologies that can effectively control particle size, shape, and crystallinity. The use of platelet or spherical morphology, as seen with LFP, ensures good slurry coating. This minimizes electrode resistance, hence improving energy density and power density. The agglomeration of powders should also be considered. Poorly dispersed powders can cause electrodes with poor uniformity, hence lowering their ionic conductivity.
Surface Coatings and Conductivity Enhancements
One of the biggest issues associated with pure LFP is that they have naturally low electronic conductivity of about 10⁻⁹ S/cm. To improve this issue of low conductivity in LFP, different methods of carbon coating are used. By coating LFP with carbon, a thin conductive layer is created around each particle. This improves electron conductivity without compromising the structural properties of the olivine framework.
Different precursors for carbon coating are used, ranging from amorphous carbon obtained by burning sugars like sucrose and glucose to sophisticated polymers and graphene. Moreover, doping of LFP with small amounts of metals like Mg²⁺, Ti⁴⁺, and Mn²⁺ has been observed to improve conductivity as well as lithium ion mobility in the crystal lattice of LFP.
High-Temperature Stability and Safety
A major advantage of LFP powder is its thermal and chemical stability. It can maintain its olivine structure at temperatures above 200 degrees Celsius, thus minimizing thermal runaway. Such batteries are thus more appropriate for use in safety-critical applications such as electric vehicle battery packs, stationary storage systems, and military-grade batteries.
In addition, LFP powders with high purity have fewer impurities such as Fe₂O₃ or lithium carbonate, which can lead to capacity loss, gas generation, or an increase in internal resistance during cycling. High-purity powders with consistent properties are thus more appropriate for use in safety-critical applications at high current densities and harsh working conditions.
Applications in High-Performance Systems
- Electric Vehicles (EVs): The rapid charge and discharge properties of advanced LFP powders are possible without compromising safety or cycle life. The carbon-coated powders are also doped with other materials, making EVs capable of providing high energy density as well as high power density.
- Grid-Scale and Renewable Energy Storage: The high-density powders with well-controlled particle morphology are used to improve the efficiency of the energy storage batteries for solar, wind, or hybrid energy storage systems.
- Industrial and Aerospace Applications: The precision-engineered powders are used for dependable energy storage in harsh environments, such as high temperature, vibration, or shock. These properties are critical for various applications in the aerospace industry.
- High Rate Consumer Electronics: Although compactness is critical for various consumer applications, the stable voltage output with long cycle life makes the use of the nanoscale carbon-coated LFP powders suitable for medical devices or power tool applications.
Future Trends in LFP Powder Technology
The development of LFP powders continues to focus on enhancing conductivity, increasing energy density, and enabling scalable, cost-effective production. Emerging strategies include:
- 3D structured LFP particles: Optimizing pore networks to enhance electrolyte penetration and lithium-ion mobility.
- Graphene and hybrid carbon coatings: Providing superior conductivity and mechanical strength.
- Continuous-flow hydrothermal synthesis: Offering scalable production of uniform, high-purity powders at lower cost.
- Recycling and circular supply chain integration: Recovering lithium and iron phosphate from end-of-life cells to produce high-performance LFP powders sustainably.
These innovations are crucial as the demand for LFP batteries surges globally, driven by EV adoption, renewable energy storage expansion, and the push for safer, longer-lasting lithium-ion technologies.
Conclusion
Advanced LiFePO₄ powder is no longer a simple precursor; it is a highly engineered material that dictates the performance, safety, and reliability of modern lithium-ion batteries. Through careful control of particle morphology, carbon coating, and elemental doping, manufacturers can produce LFP cathodes that achieve high power output, fast charging, and exceptional longevity.
Reference:
[1] Balakrishnan, N.T.M. et al. (2021). Lithium Iron Phosphate (LiFePO4) as High-Performance Cathode Material for Lithium Ion Batteries. In: Rajendran, S., Karimi-Maleh, H., Qin, J., Lichtfouse, E. (eds) Metal, Metal-Oxides and Metal Sulfides for Batteries, Fuel Cells, Solar Cells, Photocatalysis and Health Sensors. Environmental Chemistry for a Sustainable World, vol 62. Springer, Cham.